PlasmoSugars
Exploration of the singularities of the sugar nucleotide metabolism and description of novel glycosylation pathways in the malaria parasite
- Duración
- 01/01/2014 - 30/06/2017
- Coordinador
- Luis Izquierdo
- Financiadores
- MINECO (Spanish Ministry of Economy)
Glycosylation represents one of the most common and heterogeneous post-translational modifications of proteins, and complex glycoconjugates are involved in almost every biological process. Thus glycoconjugates play a major role in nearly every human disease.
Sugar nucleotides are the activated forms of sugars used as donor substrates in glycosylation reactions and our lab has precisely delineated the sugar nucleotide biosynthetic pathways in the malaria parasite. In contrast to the prevailing idea that the Plasmodium glycoconjugate structures are limited to glycosylphosphatidylinosytil (GPI) anchors, our works show that other sugar nucleotide and glycosylation pathways are active in the blood stages of the malaria parasite.
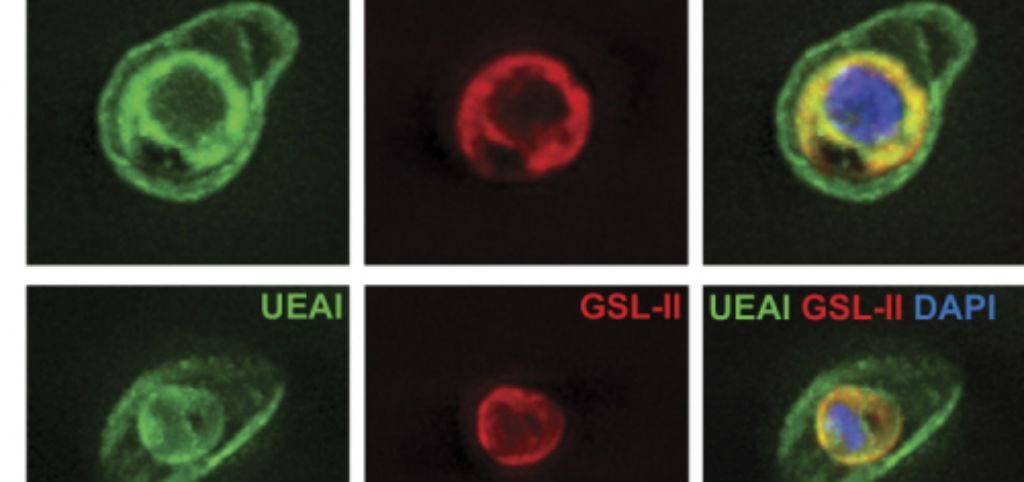
We aim to probe essential and/or unanticipated enzymatic steps in these metabolic routes that are unidentified in the P. falciparum genome. Among these unresolved issues are: i) the identity of the P. falciparum putative glucosaminephosphate N-acetyltransferase (GNA) that converts glucosamine-6-P (GlcN6P) to GlcNAc6P; ii) the source (and biological significance) of the UDP-galactose (UDP-Gal) pool detected in the blood-stages of the parasite. Furthermore, our previous studies have shown the presence of GDP-fucose (GDP-Fuc) pools through the asexual stages of P. falciparum life-cycle at levels comparable to other protozoan parasites. This, together with the incorporation of tritium labelled GDP-Fuc by cell extracts of the parasite, strongly suggests the presence of active fucosylation mechanisms in P. falciparum. Our works show that P. falciparum blood-stage cell lines with engineered alterations in the GDP-Fuc biosynthetic pathway are not labelled by the fucose-binding Ulex europaeus agglutinin I (UEA-I). Therefore, a fucosyl transferase could be involved in the synthesis of an uncharacterized UEA-I-glycotope on the surface of the parasite.
Detailed biochemical and genetic studies of these essential but much neglected biosynthetic pathways may improve our understanding of the parasite’s biology and provide urgently needed targets for chemotherapeutic intervention.
Total funding:
139,150 €
Nuestro equipo
PI
-
Luis Izquierdo Lázaro Associate Research Professor
ISGlobal team
-
 Miriam Ramirez Técnico de Soporte Científico - Laboratorio
Miriam Ramirez Técnico de Soporte Científico - Laboratorio
Otros proyectos
Ver proyectos pasadosMESA
La Alianza Científica para la Erradicación de la Malaria (MESA) tiene como objetivo avanzar en la ciencia de la erradicación de la malaria.
NHEPACHA
Nuevas Herramientas para el Diagnóstico y la Evaluación del Paciente con Enfermedad de Chagas
Estudio inmunológico de la vacuna RTS,S
Estudio de correlatos de protección frente a la malaria después de la vacunación con RTS,S/AS01E: Una evaluación inmunológica exhaustiva en el ensayo clínico de Fase III, doble ciego, aleatorizado, multicéntrico con un grupo control
Euroleish.net
Control of Leishmaniasis. From bench to bedside and community
GREPIMER
Grup de recerca en patología importada i malaties emergents i re-emergents
TESEO
Nuevos regímenes de quimioterapia y biomarcadores para la enfermedad de Chagas
ASINTMAL
Unravelling Disease Tolerance and Host Resistance in Afebrile 'P. falciparum' Infections: a Prospective Study in Mozambican Adults
ADAM
Administración masiva y focal de fármacos antimaláricos para avanzar hacia la eliminación de la malaria en Mozambique: acelerando la implementación de programas y políticas
MULTIPLY
MULTIple doses of IPTi Proposal: a Lifesaving high Yield intervention
Science4Pandemics
Citizens engagement digital platform for collective intelligence in pandemics
HIDDENVIVAX
Novel organ-on-a-chip technology to study extracellular vesicles-mediated cryptic infections in Plasmodium vivax malaria
Subclinical Infections in Children and Long Term Health Effects
Infection acquisition in early life and health outcomes in childhood - MARATO TV3
Herramienta innovadora de detección de enfermedades y vacunación a población inmigrante en riesgo en España
Project Code: PI21/00651
Impacto de las coinfecciones en el balance de respuestas de anticuerpos y linfocitos T helper a antígenos diana de inmunidad natural y vacunal frente a patógenos humanos prominentes
Project Code: PI20/00866
MENA Migrant Health
Transforming data collection and surveillance to drive migrant health research, care and policy
SexMal
Social affairs and sex in P. falciparum: implications for malaria elimination
EpiGen
Building Scalable Pathogen Genomic Epidemiology in Ethiopia
SMART
Identifying Severe Malaria with a new Aptamer-based Rapid diagnostic Test
MalTransc
Transcriptional regulation of adaptation and developmental decisions in malaria parasites: from epigenetic variation to directed transcriptional responses
GenMoz
P. falciparum genomic intelligence in Mozambique
BOHEMIA
Broad One Health Endectocide-based Malaria Intervention in Africa
RESPONSE
Mechanisms of the transcriptional responses to changes in the environment in the malaria parasite Plasmodium falciparum
VivaxEVTalk
Extracellular Vesicles as Intercellular Communicators and Biomarkers of Cryptic Erythrocytic Infections in Plasmodium vivax malaria
VaMonoS
Unravelling the heterogoneity and function of monocytes in vaccination and immunity to malaria
CLIMSOCTRYPBOL
Insight on climate and social participatory research for integral management of vectorborne zoonosis caused by Trypanosoma cruzi and Leishmania spp. in the Bolivian Gran Chaco.




