A New Bacterial Species with Pathogenic Potential is Described
The new 'Acinetobacter' species was isolated from human samples from different countries and carries antibiotic-resistant genes
16.01.2017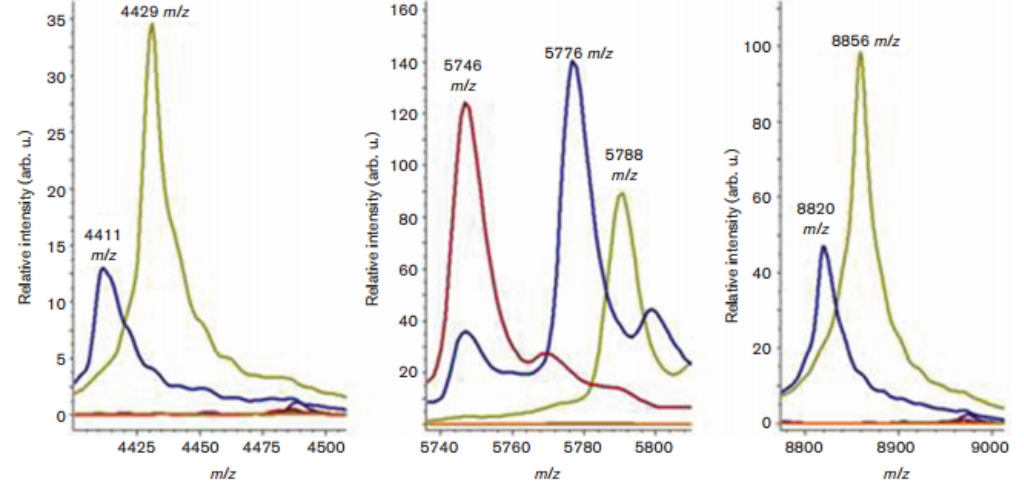
A team led by Ignasi Roca, ISGlobal researcher and Jordi Vila, director of the Antimicrobial Resistance Initiative, describes a new Acinetobacter species isolated from patient samples obtained in different countries. In a series of three recently published papers, the researchers use advanced molecular techniques to show that the isolates belong to a new species of the Acinetobacter calcoaceticus-Acinetobacter baumannii complex (ACB), and named it Acinetobacter dijkshoorniae in honor of Lenie Dijkshoorn, a dutch microbiologist that has dedicated her life to the taxonomy of the genus Acinetobacter.
Firstly, the authors characterized a group of 12 strains obtained from patients over a period of 15 years and that differed genetically from other species of the ACB complex. Using advanced molecular techniques, the authors show that these strains form a unique and distinct group, although highly related to Acinetobacter pittii. The “type strain” of the new species, baptized Acinetobacter dijkshoorniae, is JVAPo1T, a strain isolated from a Turkish patient in 2009.
In parallel, the authors sequenced the whole JVAPo1T genome and found that it carries several antibiotic-resistant genes, including the blaNDM-1 gene, that confers resistance to most beta-lactams and is localized on a plasmid (a small circular DNA molecule that can be transferred between bacteria), and the aphA6 gene, that confers resistance to some aminoglycosides. Results obtained with a bacterial pathogenicity prediction program reveal that the new species is highly likely to be a human pathogen and shares sequences with other pathogenic families, including A. baumannii strains.
Finally, the authors determined whether A. dijkshoorniae and Acinetobacter seifertii (another recently describes species) can be differentiated from other Acinetobacter species through a mass spectrometry method (MALDI-TOF/MS) that is successfully being used for the identification of other ACB species. Correct identification is important since the different Acinetobacter species differ in antibiotic susceptibility and clinical outcome. Through the use of software that processes the distinct protein profiles, the authors successfully identified species-specific signals. Inclusion of these new molecular signatures into the data base should allow the rapid and automated identification of all the Acinetobacter species within the group” explains Jordi Vila. This, in turn, will allow us to evaluate the clinical and epidemiological relevance of the different species of the family and will help improve the treatment and handling of Acinetobacter infections.”
Outbreaks of Acinetobacter infections typically occur in intensive care units and healthcare settings where very ill patients are housed. Acinetobacter is often resistant to many commonly prescribed antibiotics.
References:
Cosgaya C, Marí-Almirall M, Van Assche A, Fernández-Orth D, Mosqueda N, Telli M, Huys G, Higgins PG, Seifert H, Lievens B, Roca I, Vila J. Acinetobacter dijkshoorniae sp. nov., a member of the Acinetobacter calcoaceticus-Acinetobacter baumannii complex mainly recovered from clinical samples in different countries. Int J Syst Evol Microbiol. 2016 Oct;66(10):4105-4111.
Marí-Almirall M, Cosgaya C, Higgins PG, Van Assche A, Telli M, Huys G, Lievens B, Seifert H, Dijkshoorn L, Roca I, Vila J. MALDI-TOF/MS identification of species from the Acinetobacter baumannii (Ab) group revisited: inclusion of the novel A. seifertii and A. dijkshoorniae species. Clin Microbiol Infect. 2016 Dec 3.
Fernández-Orth D, Cosgaya C, Telli M, Mosqueda N, Marí-Almirall M, Roca I, Vila J. Draft Genome Sequence of JVAP01T, the Type Strain of the Novel Species Acinetobacter dijkshoorniae. Genome Announc. 2017 Jan 12;5(2).



